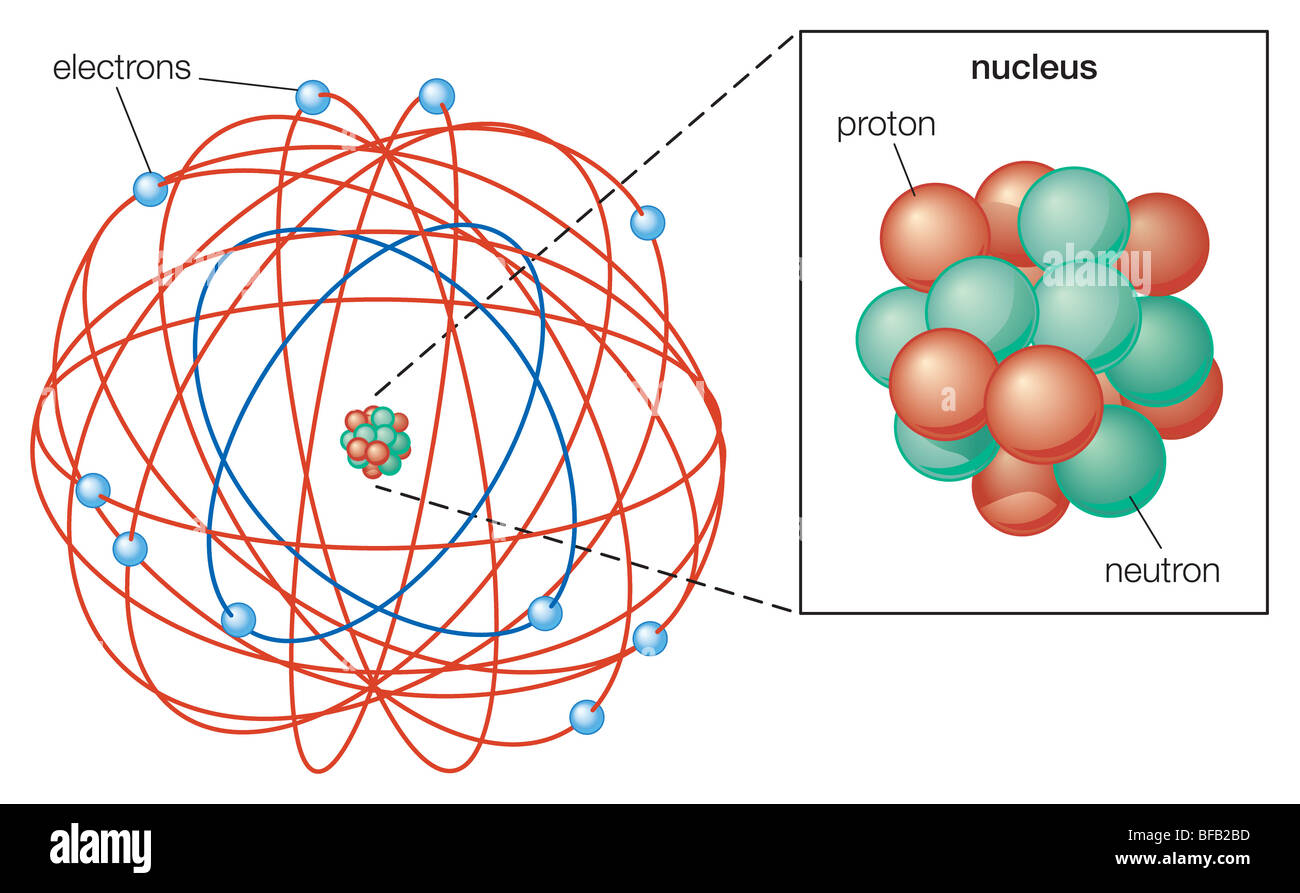
A positively-charged centre in an atom is called the nucleus.Compared to the size of an atom, the size of the nucleus is tiny.īased on his experiment, Rutherford put forward the model of an atom, which had the following features:.The electrons revolve in orbit around the nucleus in circular paths.The positively-charged centre of an atom is the nucleus that resides in an atom’s whole mass.Rutherford concluded the following through his experiment: But even then, some particles deflected at HUGE angles. The further they moved around the screen, the fewer particles were found. Most of the alpha particles went straight through the gold foil as if it weren’t even there and hit the screen at an angle of zero degrees.  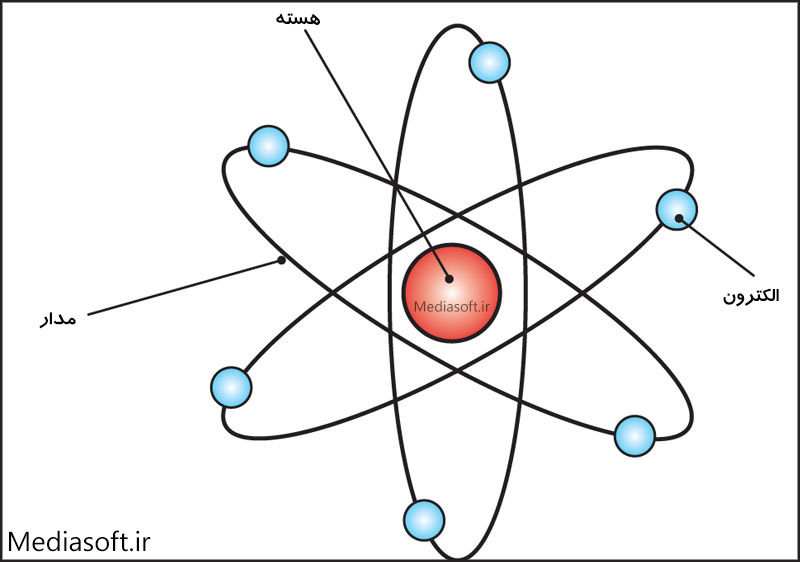
After the experiment, we understood that the positive charge was concentrated in the nucleus and that most of the atom is space. This was called the plum pudding model because the negative charges were like plums stuck in the positively-charged plum pudding.īut Rutherford’s experiment, also known as the Geiger-Marsden experiment, changed everything. Before his famous experiment with gold foil, scientists imagined the atom as a large area of positive charge, with negative charges stuck on the outside. To study the deflection of alpha particles, he placed a fluorescent screen coated with ZnS.Įrnest Rutherford was a British physicist who is particularly famous for studying the structure of the atom. In his investigation, he directed high energy streams of alpha particles from a radioactive source at a thin sheet(100 nm thickness) of gold. J.J Thomson was the first scientist who proposed the atom model, considered a ‘Plum pudding model.’ Later on, Rutherford introduced a new model widely accepted and considered the ‘ Nuclear model of the atom.’ Rutherford had shown his model with the help of an experiment. He realised this because most of the alpha particles passed straight through the piece of gold foil, with just a few deflected at huge angles. Rutherford’s gold foil experiment showed that atoms are mostly empty space, with the positive charge concentrated in a nucleus.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
